Dear colleagues and group members,
Welcome to my monthly update on medical device regulations in Russia and countries of the Eurasian Economic Union (EEU). Here are my three highlights for May 2016:
- Development of the Eurasian Medical Device Regulations
In May 2016, the Eurasian commission published final approved versions of five second-level regulations (which were previously available as drafts) for the Agreement on Common Principles and Rules of Circulation of Medical Devices in the Eurasian Economic Union (EEU):
- Essential principles and rules on the safety and effectiveness of medical devices (link in Russian) – this document was drawn up on the basis of similar GHTF guidance and provides general and fundamental design and manufacturing requirements on the safety, quality and performance of medical devices (MD) and in-vitro medical devices (IVD). In addition, this document provides common labelling requirements for MD and IVD within the Eurasian Economic Union
- Rules on conducting technical testing for medical devices (link in Russian) and Rules on conducting clinical trials (link in Russian) provide guidance on conducting pre-submission testing and trials. Good news for IVD manufacturers – in contrast to the current Russian approval process, only clinical trials will be needed in the EEU (skipping the technical tests). Not so good news for implantable devices – full-scale clinical trials with patients’ involvement will be mandatory for their approval
- Resolution on the EEU medical device mark (link in Russian) – this document provides detailed descriptions of new EAC (EEU) medical device symbols and procedures of EAC-marking for medical devices approved under the new harmonised approval process
- Resolution on the formation and maintenance of the common medical devices information system (link in Russian), which describes procedures for the common Eurasian database of approved medical devices, clinical centres and laboratories accredited to conduct trials/testing and information on medical devices’ adverse reactions and safety field actions within the EEU.
In these ways, today the full Eurasian medical device regulatory model can be vizualized like this:
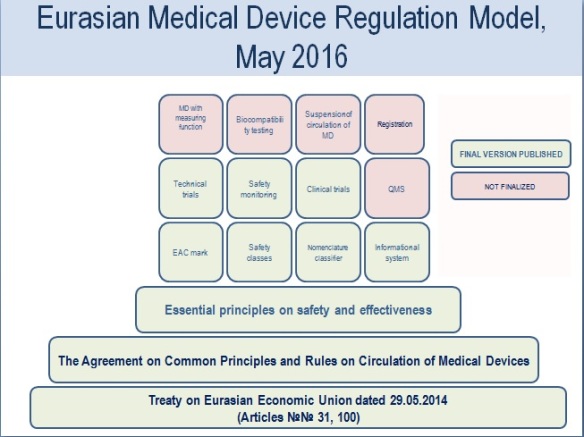
Although currently the new EEU procedures do not work in practice, according to the Eurasian commission, a full-scale launch of the new harmonised system is expected in 2016.
- The Russian Ministry of Health is considering an extended list of Implantable Medical Devices
The Russian Ministry of Health is preparing an extension of the “List of Implantable Medical Devices for Medical Aid Provided to Citizens under the Programme of State Guarantees of Free Medical Care“. According to information published in May 2016 (link in Russian), the Russian regulator reviewed 671 applications from medical device manufacturers and medical institutions, with positive decisions made on the majority of them.In this context, it should be recalled that the last version of the list (with 207 items) was published in December 2014. According to the Russian Ministry of Health, companies producing Implantable Medical Devices not included in the list will not able to participate in national healthcare programmes from 2017. According to current procedures, applications for inclusion in the list can be submitted to the Ministry of Health by 1st March each year.
- New Version of Guidelines for Expertise in Medical Devices
A draft of new guidelines on the expertise in medical devices for procedures of registration was published in May 2016 (link in Russian). This document contains detailed descriptions of the expertise procedure, requirements on documentation format for registration and testing files, and unifies these requirements from two expert organisations. The document will replace the previous version of these guidelines (link in Russian) published in November 2013 and will be expected to come into force formally in the near future.
Thank you for following the blog and newsletter. I am always grateful for your comments and encourage you to ask specific questions in the LinkedIn group.
