Dear Colleagues,
Here is my regular update on the Russian and Eurasian medical device regulations for the last month:
- Update on the Eurasian Transitional Period
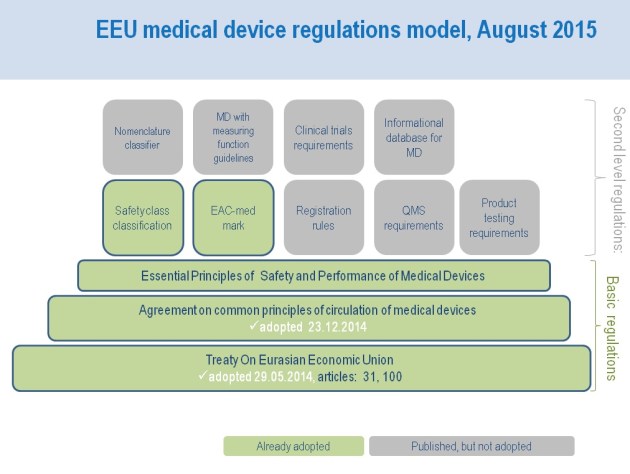
On 9 March 2021, the Eurasian Commission published Regulation No.28 (Link in Russian) which confirmed and specified deadlines for the Eurasian regulation transition period previously established as follows:
- Submissions for registration and expertise of medical devices according to the local rules (of the Eurasian member states) will be open until 31 December 2021.
- If submitted before 31 December 2021, the registration dossier will be reviewed according to the local registration rules of a member state.
- Medical devices approved according to the local registration system in the Eurasian member states may stay approved in this member state by the end of validity of the registration.
- Registration certificates for medical devices registered according to the local rules may be amended (by the local rules) by 31 December 2026.
On 22 March 2021, the Russian Ministry of Health published the draft of amendments of the rules for the state registration of medical devices in Russia (link to the draft in Russian), harmonised with Eurasian regulations and transition deadlines and proposed other amendments. The draft is currently in the discussion stage until 9 April 2021.
Regardless of the approaching end of the transition period, we still may see a limited number of submissions and approvals according to the new Eurasian system. As of the end of March 2021, there are five records concerning approval of medical/in-vitro devices in the Eurasian Union. All approved devices are manufactured by Eurasian manufacturers.
- Extension of the Restriction List for Medical Devices in Russia
On 6 March 2021, the Russian Government released Resolution No.336 (link in Russian), which expanded the list of medical devices originating from foreign countries, subject to restrictions for state and municipal procurements according to Resolution #102.
The new devices added to the restriction list include biochemical analysers, fetal monitors, medical, biological, and clinical microscopes for laboratory diagnostics, ultrasound surgical devices, surgical coagulants, and therapeutic laser devices.
According to Resolution #102, participation in government and municipal tenders for the supply of these products must be rejected if at least two suppliers from Russia or the EAEU countries take part in the auction with a similar type of product.
- Kazakhstan Updated Labelling and Advertising Requirements for Pharmaceutical Products and Medical Devices
At the end of February 2021, the Ministry of Health of Kazakhstan released order #KR-DCM -11 (link in Russian) and updated national requirements for the labelling of medical devices. The regulations provide a list of information and symbols required for the manufacturer to state on the label of a medical device. The labelling must be present in Russian and Kazak languages, approved by the competent authority during the registration process and attached to each unit.
Previously, in January 2021, the Ministry of Health of Kazakhstan enforced the order #KR-DCM -11 (link in Russian) and updated rules for advertising medical devices in the country.
*****
Thank you for following and for your continued reading of my blog. Let me remind you that you can subscribe to my emails via the ‘follow’ button on the toolbar of this blog.